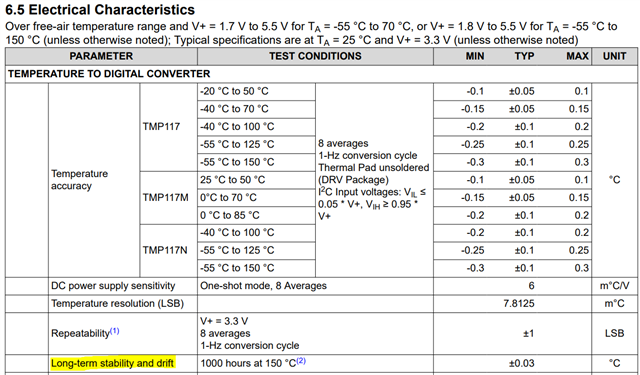
We are using the TMP117 temperature sensors in one of our medical device tester setups. We need a tolerance of 0.1 degree Celsius over the range of 31 to 42 degrees Celsius. Technically this part satisfies our needs and proved to be acceptable upon testing. But since this is for testing medical devices, we want to keep a track of its calibration. The following are a couple of questions I have. Please answer them as you see fit. Thank you!
1. Are the initial NIST traceable calibration records accessible upon request? Are these traceable by lot or should this information be requested before purchasing?
2. Given that we do not reprogram its EEPROM, what is the recommended next calibration date? Should this be calibrated once every year? Please point me to the right documentation for this.
3. (Optional) If there is another TI product that best suits our needs of calibration traceability, please point me to it. If not, we have been quite satisfied with TMP117 so far.
Again, thank you for your support!